Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Abe, Toru*; Hirano, Fumio; Mihara, Morihiro; Honda, Akira
Genshiryoku Bakkuendo Kenkyu (CD-ROM), 27(1), p.3 - 11, 2020/06
Degradation of TRU waste in a geological disposal facility may cause the formation of a nitrate plume. A Nitrate Evolution model due to mineral reactions, microbial activity, and metal corrosiON (NEON) has therefore been developed to evaluate the safety case for geological disposal of TRU waste. Small scale laboratory experiments can be reproduced satisfactorily, however, it is necessary to demonstrate the applicability of the NEON model on scales relevant to the geological disposal of TRU waste. In the current study, an industrial analogue of a nitrate plume from the pollution of groundwater from nitrate fertilizers used on Ikuchi Island, Japan was selected to test the applicability of the NEON model. Concentration profiles of nitrate ions in the groundwater were successfully reproduced over the hundreds of meters scale demonstrating the applicability of the NEON model in evaluating the chemical behavior of a nitrate plume derived from the geological disposal of TRU waste.
Kaneko, Masashi; Watanabe, Masayuki; Matsumura, Tatsuro
Dalton Transactions, 45(43), p.17530 - 17537, 2016/11
Times Cited Count:33 Percentile:87.63(Chemistry, Inorganic & Nuclear)Relativistic density functional calculations were applied to study the separation behaviors of Am(III) ion from Eu(III) ion by diglycolamide (DGA) and nitrilotriacetamide (NTA) ligands in order to understand the difference in the separation mechanism of their reagents. The complexation reaction was modeled on the basis of previous experimental studies. The calculated energies based on stabilization by complex formation at the ZORA-B2PLYP/SARC level predicted that the DGA reagent preferably coordinated to Eu(III) ion when compared with Am(III) ion. In contrast, the NTA reagent selectively coordinated to Am(III) ion when compared with Eu(III) ion. These results reproduced the experimental selectivity of DGA and NTA ligands toward Eu(III) and Am(III) ions. Mulliken's population analyses implied that the difference in the contribution of the bonding property between the f-orbital of Am and donor atoms determined the comparative stability of Eu and Am complexes.
Nakamoto, Tatsushi*; Sugano, Michinaka*; Xu, Q.*; Kawamata, Hiroshi*; Enomoto, Shun*; Higashi, Norio*; Idesaki, Akira; Iio, Masami*; Ikemoto, Yukio*; Iwasaki, Ruri*; et al.
IEEE Transactions on Applied Superconductivity, 25(3), p.4000505_1 - 4000505_5, 2015/06
Times Cited Count:0 Percentile:0(Engineering, Electrical & Electronic)Recently, development of superconducting magnet system with high radiation resistance has been demanded for application in accelerator facilities such as CERN LHC. In order to realize superconducting magnet system with high radiation resistance, it is necessary to develop electrical insulator with high radiation resistance because the electrical insulator is made of organic materials whose radiation resistance is inferior to that of inorganic materials. We developed a glass fiber reinforced plastic with bismaleimide-triazine resin. The developed material showed excellent radiation resistance; the material evolved gases of 5 10
10  mol/g and maintained flexural strength of 640MPa (90% of initial value).
mol/g and maintained flexural strength of 640MPa (90% of initial value).
Nagasawa, Naotsugu
Nihon Setchaku Gakkai-Shi, 41(10), p.420 - 426, 2005/10
no abstracts in English
Nagasawa, Naotsugu; Kaneda, Ayako*; Kanazawa, Shinichi*; Yagi, Toshiaki; Mitomo, Hiroshi*; Yoshii, Fumio; Tamada, Masao
Nuclear Instruments and Methods in Physics Research B, 236(1-4), p.611 - 616, 2005/07
Times Cited Count:80 Percentile:97.88(Instruments & Instrumentation)no abstracts in English
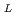 -lactic acid) by radiation-induced crosslinking
-lactic acid) by radiation-induced crosslinkingMitomo, Hiroshi*; Kaneda, Ayako*; Tran, M. Q.*; Nagasawa, Naotsugu; Yoshii, Fumio
Polymer, 46(13), p.4695 - 4703, 2005/06
Times Cited Count:116 Percentile:94.31(Polymer Science)no abstracts in English
Nagasawa, Naotsugu; Yagi, Toshiaki
Science & Technology Journal, 13(10), p.20 - 21, 2004/10
no abstracts in English
Kono, Hidetoshi; Saven, J. G.*
Seibutsu Butsuri, 43(4), p.186 - 191, 2003/07
no abstracts in English
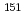 Eu M
Eu M ssbauer spectroscopic study on the Eu
ssbauer spectroscopic study on the Eu M
M O
O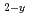 (0
(0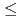 y
y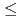 1.0)(M=Th, U)
1.0)(M=Th, U)Masaki, Nobuyuki; Otobe, Haruyoshi; Nakamura, Akio; Harada, Daijitsu*; Ito, Kentaro*; Sasaki, Yoshinobu*; Hinatsu, Yukio*
Journal of Nuclear Science and Technology, 39(Suppl.3), p.217 - 220, 2002/11
no abstracts in English
Kuramoto, Kenichi; Yamashita, Toshiyuki; Shiratori, Tetsuo
Progress in Nuclear Energy, 38(3-4), p.423 - 426, 2001/02
Times Cited Count:6 Percentile:44.09(Nuclear Science & Technology)no abstracts in English
Nakano, Yoshihiro; Ando, Masaki; Okajima, Shigeaki; Takano, Hideki; Akie, Hiroshi
Progress in Nuclear Energy, 38(3-4), p.343 - 346, 2001/02
Times Cited Count:1 Percentile:12.01(Nuclear Science & Technology)no abstracts in English
Hidaka, Akihide; Nakamura, Takehiko; Nishino, Yasuharu; Kanazawa, Hiroyuki; Hashimoto, Kazuichiro; Harada, Yuhei; Kudo, Tamotsu; Uetsuka, Hiroshi; Sugimoto, Jun
JAERI-Conf 99-005, p.211 - 218, 1999/07
no abstracts in English
Bamba, Tsunetaka; Mitamura, Hisayoshi; ; *;
JAERI-Review 98-002, 23 Pages, 1998/02
no abstracts in English
Hirata, Masaru; Sekine, Rika*; Onoe, Jun*; Nakamatsu, Hirohide*; Mukoyama, Takeshi*; *; Tachimori, Shoichi
Journal of Alloys and Compounds, 271-273, p.128 - 132, 1998/00
Times Cited Count:8 Percentile:52.63(Chemistry, Physical)no abstracts in English
Shirai, Toshizo; Sugar, J.*; Wiese, W. L.*
JAERI-Data/Code 97-031, 115 Pages, 1997/07
no abstracts in English
Shirai, Toshizo; Sugar, J.*; Wiese, W. L.*
JAERI-Data/Code 97-030, 63 Pages, 1997/07
no abstracts in English
Shirai, Toshizo; Sugar, J.*; Wiese, W. L.*
JAERI-Data/Code 97-029, 60 Pages, 1997/07
no abstracts in English
Shirai, Toshizo; Sugar, J.*; Wiese, W. L.*
JAERI-Data/Code 97-028, 72 Pages, 1997/07
no abstracts in English
Shirai, Toshizo; Sugar, J.*; Wiese, W. L.*
JAERI-Data/Code 97-027, 75 Pages, 1997/07
no abstracts in English
Shirai, Toshizo; Sugar, J.*; Wiese, W. L.*
JAERI-Data/Code 97-026, 136 Pages, 1997/07
no abstracts in English